Electronic Data Capture (EDC) software is a vital tool for managing and organizing data in fields such as clinical research. It simplifies the process of collecting, storing, and analyzing data, making it indispensable in industries like healthcare and pharmaceuticals. Selecting the right EDC software is crucial for ensuring the success of research projects. In this article, we’ll delve into the world of the best electronic data capture (EDC) software, discussing the top options and highlighting the features that set them apart in the realm of electronic data capture.
What we cover
Benefits & Applications
Electronic Data Capture (EDC) software offers several advantages, including enhanced data accuracy and completeness, increased efficiency through automation, improved data security and compliance, real-time data analysis, and enhanced collaboration among stakeholders.
EDC is primarily used in clinical trials, pharmacovigilance, epidemiology, and medical research by organizations like pharmaceutical companies, clinical research firms, academic institutions, government agencies, and non-profits. It collects data on symptoms, side effects, medication adherence, medical history, lifestyle, adverse drug reactions, public health surveys, and research studies on new drugs and medical devices.
In essence, EDC software is an essential tool that elevates data quality, efficiency, and security in various industries and research settings.
What are data sources?
Data sources are crucial for Electronic Data Capture software, serving as the reservoirs from which data is gathered. EDC software draws data from various sources, both internal and external, to support clinical trials and research studies effectively. These sources include electronic health records (EHRs), clinical trial management systems (CTMSs), laboratory information systems (LISs), patient-reported outcome (PRO) systems, and wearable devices.
Data sources play a pivotal role in EDC software by providing the data necessary to support clinical trials or research studies. Through automation, EDC software enhances the efficiency and accuracy of data collection, ultimately leading to more dependable results and informed decision-making.
General Pricing
The cost of EDC software varies based on factors such as study size, complexity, the number of users, and required features. On average, EDC software typically falls within a range of $600 to $4,000 per month. For a 24-month study, this could mean a total cost of $14,400 to $96,000. It’s essential to carefully consider these factors and budget constraints when selecting EDC software.
The pricing of EDC software is influenced by factors like study size and complexity, the number of users, required features, deployment model (on-premise or cloud), and the choice of vendor. Making the right choice requires organizations to assess their specific needs and budget carefully.
Choosing the right EDC Software
Selecting the best Electronic Data Capture (EDC) software is a critical decision for organizations involved in clinical trials, research, and data management. To make an informed choice and avoid costly mistakes, here are several key factors to consider:
- Integration Compatibility: Ensure the software works well with your existing systems.
- Security and Compliance: Check for strong data security and regulatory compliance features.
- Usage and Scalability: Look for user-friendly software that can grow with your needs.
- Data Collection and Management: Assess data capture, management, and reporting capabilities.
- Support and Training: Consider vendor support and training options.
- Cost and Budget: Evaluate the software’s cost in relation to its value and your budget.
selecting the right EDC software involves a thorough evaluation of integration capabilities, security, compliance, user-friendliness, scalability, data management features, support, and cost considerations. By carefully weighing these factors, organizations can make an informed decision that minimizes the risk of choosing the wrong software and ensures the successful management of clinical trial and research data.
Best Electronic Data Capture (EDC) Software
Let’s look at a curated list of the best software options available. These selections cover a range of categories and are chosen for their outstanding features and capabilities. Whether you’re in search of productivity tools, design software, or any other type of application, our list has you covered.
Best Electronic Data Capture (EDC) Software – At a Glance
| Features | RedCap | Viedoc EDC | Grooper | Medrio |
| Best For (Features) | Research & Data Collection | Clinical Trials | Document Capture | Clinical Research |
| Ease of Use | Moderate | User-Friendly | Moderate | User-Friendly |
| Popularity | Widely Used | Growing | Emerging | Growing |
| Reviews | Positive | Positive | Positive | Positive |
| Price | Free to Low-Cost | Contact Vendor | Contact Vendor | Contact Vendor |
| Software (iOS or Windows) | Web-Based | Web-Based | Windows | Web-Based |
| Data Security | Strong | Robust | Varies | Strong |
| Customization | Highly Configurable | Configurable | Yes | Highly Configurable |
| Integration Compatibility | Works with Various Systems | Extensive | Limited | Extensive |
| Data Validation | Limited | Advanced Edit Checks | Advanced OCR | Advanced Validation |
| Data Monitoring | Limited | Real-Time Monitoring | N/A | Real-Time Monitoring |
| User Support | Online Resources | 24/7 Customer Support | Contact Vendor | Dedicated Support |
| Automation | Limited | Limited | Workflow Automation | Limited |
| Document Classification | N/A | N/A | Advanced OCR | N/A |
| Data Extraction | N/A | N/A | Data Capture and Extraction | N/A |
Best Electronic Data Capture (EDC) Software – Let’s Dive Deeper
Let’s checkout the best electronic data capture (EDC) software now!
1. RedCap
RedCap is a widely-used, user-friendly EDC system best suited for research and data collection.

Key Features:
- Versatile data collection
- Integration compatibility
- Data security
Ratings: 4.6
Pros:
- Free to low-cost
- Web-based
- Strong data security
Cons: Moderately user-friendly.
Pricing:
- Free
- Custom Pricing
RedCap stands out for its user-friendly interface, ensuring that even those without programming skills can effortlessly create and manage surveys and databases. It offers an array of powerful features, from intricate survey designs to robust data handling and report generation. Crucially, RedCap is a secure platform, safeguarding user data from unauthorized access.
RedCap simplifies survey creation, allowing for the design of complex surveys with various question types and advanced features like branching logic and skip logic. It empowers researchers to collect high-quality data efficiently.
RedCap excels in data management, offering tools to import and export data, perform data cleaning, and create data dictionaries. It even provides statistical analysis tools, making it a comprehensive solution for researchers.
Verdict: It’s an ideal choice for researchers at universities, hospitals, and other organizations, as well as businesses looking to gather data from customers or employees.
2. Viedoc EDC
Viedoc EDC is a user-friendly EDC system tailored for clinical trials.

Key Features:
- Electronic data capture
- Clinical trial management
- Data analysis and reporting
Ratings: 4.5
Pros:
- Growing popularity
- Real-time monitoring
- 24/7 customer support.
Cons: The software can be slow at times according to some users
Pricing: Custom Pricing
Viedoc EDC is a top choice for clinical trial sponsors and CROs due to its user-friendly interface, robust capabilities, and affordability. It simplifies the clinical trial process, even for users without prior EDC software experience. Viedoc EDC offers features like electronic data capture, trial management, data analysis, and reporting, making it a comprehensive solution.
This versatile platform handles a wide range of trials, from small Phase I to extensive Phase III studies, including complex trials with multiple arms and endpoints. It excels in electronic data capture, supporting various data types and forms like eCRFs, ePROs, and eConsent.
Viedoc EDC streamlines trial management tasks like subject scheduling and randomization, and it integrates well with other systems. It also provides robust data analysis tools with real-time reporting and data visualization, and it integrates smoothly with statistical analysis software like SAS and SPSS.
Look at this easy setup video using Viedoc!
Verdict: It’s an excellent choice for organizations looking for an accessible yet powerful clinical trial management solution.
3. Grooper
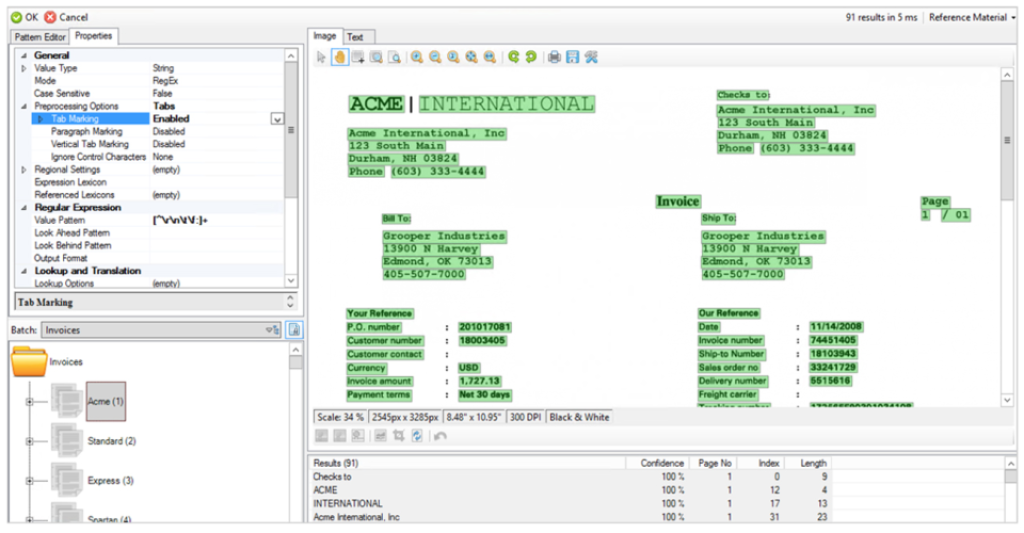
Grooper is a document capture software with advanced OCR capabilities.
Key Features:
- Advanced OCR
- Workflow automation
- Document classification
Ratings: 4.5
Pros: Best for document capture and data extraction.
Cons:
- Complexity for some users
- Pricing details not disclosed
Pricing: Custom Pricing
Grooper, a widely-used document processing software, is praised for its precision, adaptability, and scalability. Businesses of all sizes rely on Grooper to automate tasks like processing invoices, contracts, and customer onboarding.
Grooper excels in extracting data from various document types, including PDFs, Word documents, and Excel spreadsheets. It’s proficient at classifying documents, identifying critical data points, and routing documents for processing. Key strengths include:
Grooper is highly accurate in data extraction, employing machine learning and AI for consistent results. It’s a versatile software that can be tailored to meet the unique needs of any business. Grooper efficiently manages large document volumes, making it suitable for businesses of all sizes.
Verdict: Grooper is ideal for businesses, small or large, seeking to automate document processing, especially when dealing with substantial document volumes like invoices, contracts, and customer onboarding forms.
4. Fluix
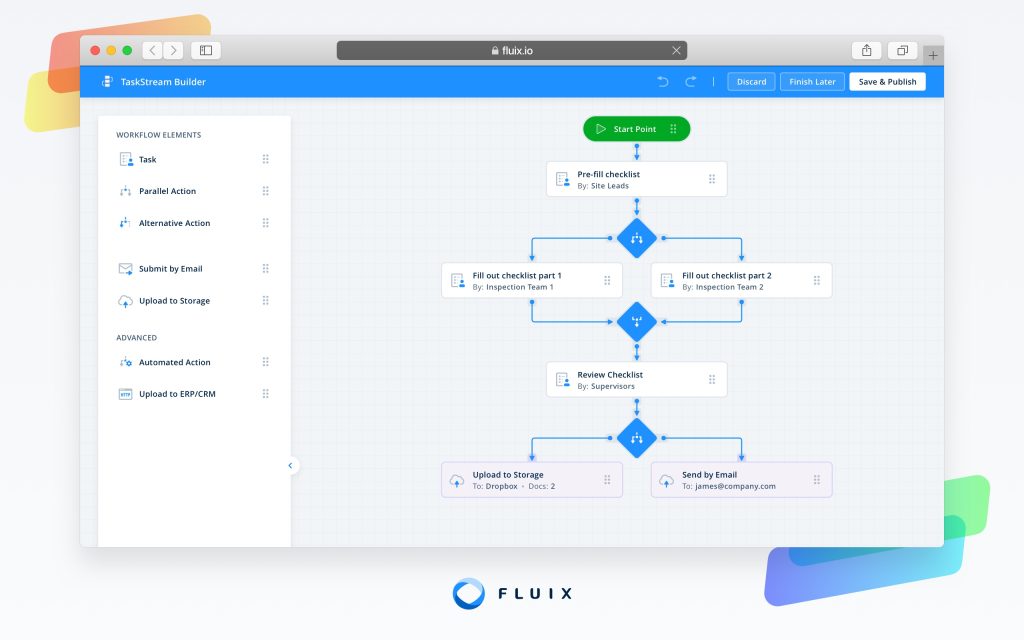
Fluix is a mobile data collection software designed for ease of use.
Key Features:
- iOS and Android compatibility
- Real-time data synchronization
- Offline access
- Mobile-friendly interface
Ratings: 4.8
Pros:
- Easy to use interface
- Wide range of features
- Affordable pricing
Cons:
- Limited data validation
- Pricing details not disclosed
Pricing: $30 per user per month
Fluix, a cloud-based mobile field service management software, is favored for its user-friendly nature, robust capabilities, and cost-effectiveness. Users appreciate its simplicity in creating forms, data collection, and report generation, along with features like electronic forms, digital signatures, and real-time data syncing. Moreover, its affordability appeals to businesses of various sizes.
Fluix is versatile, finding applications in construction, field service, and inspections. It excels in tasks like form management, data collection, report generation, workflow management, and securing digital signatures. Notable features include real-time data synchronization, offline access, integration with other systems, and a mobile-friendly interface.
For an affordable, powerful, and user-friendly mobile field service management software, Fluix is an excellent choice. It’s employed by businesses across industries and trusted by industry leaders.
Verdict: Fluix suits businesses of all sizes seeking automation in field service operations, especially in construction, field service, and inspections.
5. iMednet
iMednet is a user-friendly EDC system tailored for clinical data management.
Key Features:
- Ease of use
- Scalability
- Integration with other clinical trial management systems and EHRs
Ratings: 5
Pros: Established platform, robust data monitoring, dedicated support.
Cons: Pricing details not publicly disclosed.
Pricing: Custom Pricing
Users are drawn to iMednet for its comprehensive and accessible nature. The CTMS and EDC software it offers cover a wide spectrum of critical features. This includes clinical trial management components like subject oversight, randomization, scheduling, event adjudication, inventory control, and reporting.
Moreover, electronic data capture elements such as eCRFs, ePROs, and mobile data capture, are also integrated. Users find iMednet’s interface user-friendly and it offers the scalability needed to cater to clinical trials of varying sizes.
iMednet’s software is adaptable, finding its utility across a diverse range of clinical trials, from smaller Phase I initiatives to extensive Phase III studies. This versatility extends to therapeutic areas such as oncology, cardiology, and infectious diseases.
Verdict: It caters to the requirements of clinical trial sponsors, CROs, and research organizations of varied sizes, garnering trust within the research community. iMednet offers a powerful means to enhance clinical trial management and research acceleration.
6. ClinVigilant
ClinVigilant specializes in data monitoring and safety for clinical research.

Key Features:
- Data monitoring and safety
- Real-time alerting
- Advanced reporting
Ratings: 4.5
Pros:
- Focused expertise in data safety
- Real-time alerting
Cons: Pricing available upon request.
Pricing: Custom Pricing.
ClinVigilant is a pioneering decentralized clinical trial platform driven by blockchain technology, streamlining the entire clinical trial process for patients, research sites, and sponsors. The platform offers remote trial management and patient-site interactions via video and text chats.
It employs blockchain for creating an audit trail of interactions, ensuring transparency and accountability. ClinVigilant’s adaptable, web-based, iPad, and mobile app solutions are compatible with both Android and iOS. Furthermore, it seamlessly integrates with patient-owned and wearable devices, enhancing the efficiency of clinical trials.
ClinVigilant’s appeal lies in its decentralization, providing enhanced security and resilience against cyber threats. Its use of blockchain ensures complete transparency with a comprehensive audit trail.
Verdict: the platform facilitates remote trial conduct, reducing patient travel and participation barriers. Its modular and customizable products make it adaptable to various clinical trial needs, enhancing flexibility and efficiency.
7. Medrio
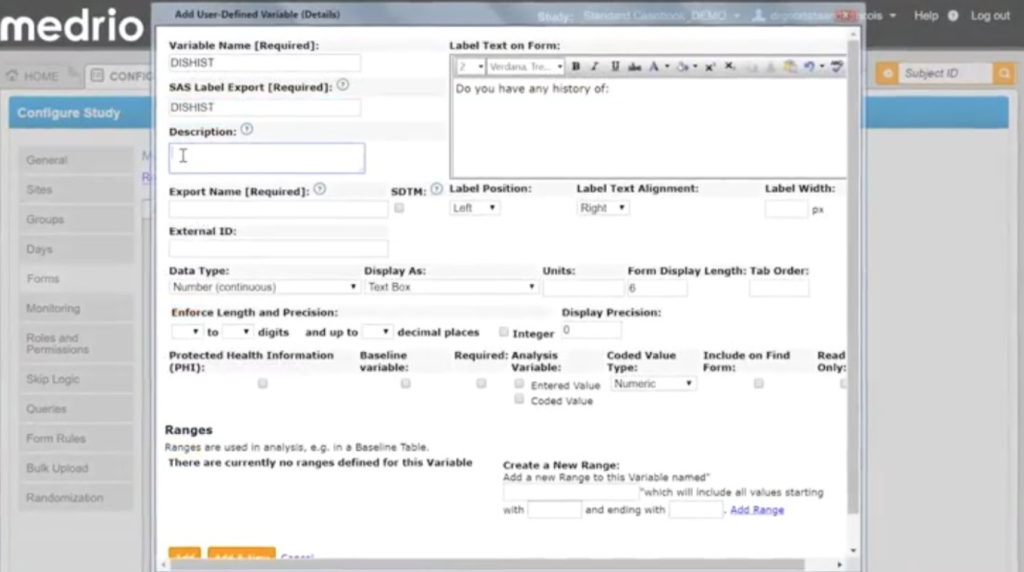
Medrio is a versatile EDC system designed for clinical research.
Key Features:
- Data capture
- User-friendly interface
- Real-time monitoring
Ratings: 4.6
Pros:
- User-friendly EDC,
- Real-time monitoring
Cons: Pricing details not publicly disclosed.
Pricing: Custom Pricing
What attracts users to Medrio’s software is its user-friendliness, robust functionality, and adaptability. Their EDC and CTMS software offer a wide array of capabilities, encompassing electronic data capture, clinical trial management, and data analysis. These features empower users to efficiently manage all aspects of their clinical trials.
Medrio’s software is versatile, making it suitable for a broad spectrum of clinical trials, regardless of their scale or therapeutic focus. It effectively handles everything from small Phase I trials to extensive Phase III studies across various medical domains, including oncology, cardiology, and infectious diseases.
The strength of Medrio’s software lies in its comprehensiveness, user-friendly interface, scalability, and seamless integration with other clinical trial management systems and electronic health records (EHRs). It’s a go-to choice for organizations seeking a powerful yet easy-to-use solution to streamline their clinical trial management.
Verdict: If you’re in the market for clinical trial management software that is not only robust but also easy to navigate and adaptable, Medrio is a compelling option to consider.
8. Castor EDC
Castor EDC is a web-based platform that excels in data capture for research purposes.
Key Features:
- Data capture
- Integration compatibility
- User-friendly
Ratings: 4.7
Pros:
- Web-based
- Seamless data capture
- Strong integration.
Cons: Pricing details not publicly disclosed.
Pricing: Personalized Pricing
Castor EDC is a cloud-based EDC platform favored for its adaptability, scalability, and user-friendliness. It caters to both clinical trial sponsors and CROs, making data collection and management more efficient.
Users appreciate Castor EDC for its flexibility, scalability, and user-friendly interface. It suits a wide range of clinical trials, from small Phase I to extensive Phase III studies.
Castor EDC stands out for its adaptability in managing diverse trials, scalability to accommodate various trial sizes, and an intuitive interface. It offers robust data management tools, including data cleaning, validation, and analysis. Additionally, it provides real-time and customized reporting.
Verdict: For those seeking an EDC platform that combines flexibility, scalability, and ease of use, Castor EDC is an ideal choice.
9. Oracle Clinical
Oracle Clinical is a comprehensive EDC system designed for clinical trials.
Key Features:
- Clinical trials support
- Data management
- Advanced reporting
Ratings: 4.1
Pros:
- Comprehensive platform for clinical trials
- Robust data management
Cons: Pricing available upon request.
Pricing: Custom Pricing
Oracle Clinical is a robust suite of clinical trial management software (CTMS) and electronic data capture (EDC) solutions trusted by clinical trial sponsors and contract research organizations (CROs). It provides end-to-end support for all phases of clinical trials.
The CTMS and EDC software within Oracle Clinical offer a diverse range of capabilities, encompassing clinical trial management, electronic data capture, data management, and reporting. Its real-time data analysis and reporting capabilities stand out as particularly powerful features.
Oracle Clinical is renowned for its adaptability and scalability. It can be customized to meet the unique requirements of each clinical trial, making it a versatile choice for trials of various sizes and complexities. Furthermore, it integrates seamlessly with other clinical trial management systems and electronic health records (EHRs).
Verdict: If you’re on the hunt for a comprehensive, potent, and flexible solution for clinical trial management, Oracle Clinical is a compelling choice. It caters to clinical trial sponsors and CROs of all sizes across various therapeutic areas.
10. Open Clinica
Open Clinica is an established EDC system well-suited for clinical research projects.
Key Features:
- Clinical research support
- Data capture
- User-friendly interface
Ratings: 4.6
Pros:
- Established platform for clinical research
- User-friendly interface
Cons: Pricing details not publicly disclosed.
Pricing: Custom Pricing
OpenClinica offers open-source clinical trial management software (CTMS) and electronic data capture (EDC) solutions for diverse users, including clinical trial sponsors, contract research organizations (CROs), and academic institutions. It efficiently manages all aspects of clinical trials.
OpenClinica’s appeal lies in its open-source, powerful, and flexible nature. Its software features include clinical trial management, electronic data capture, data management, and reporting, with a focus on real-time data analysis and reporting.
OpenClinica’s open-source nature makes it a cost-effective choice for clinical trials, irrespective of budget. Its adaptability and scalability suit trials of various sizes and complexities. Additionally, a robust user and developer community provides rapid support.
Verdict: For those seeking a versatile, budget-friendly solution for clinical trial management, OpenClinica fits the bill. It serves a wide user base and various therapeutic areas, offering efficient tools for remote data collection and tailored research needs.
FAQs
What are best edc systems for clinical trials?
Selecting the right EDC system for clinical trials depends on specific needs. Top EDC systems include Medidata Rave EDC, Oracle Clinical, Veeva Vault Clinical, Castor EDC, CRF Health ClinTrial, and OpenClinica EDC. The choice should align with the trial’s requirements.
Is Electronic Data Capture (EDC) Software Easy To Learn?
Learning EDC software is feasible, especially with prior computer experience. Training and online resources are available to help users grasp tasks like creating eCRFs, data entry, and reporting. To learn EDC software effectively, users should take vendor-provided training, use online resources, practice in a non-production environment, and seek assistance when needed. Despite its complexity, EDC software is learnable with training and practice.








